
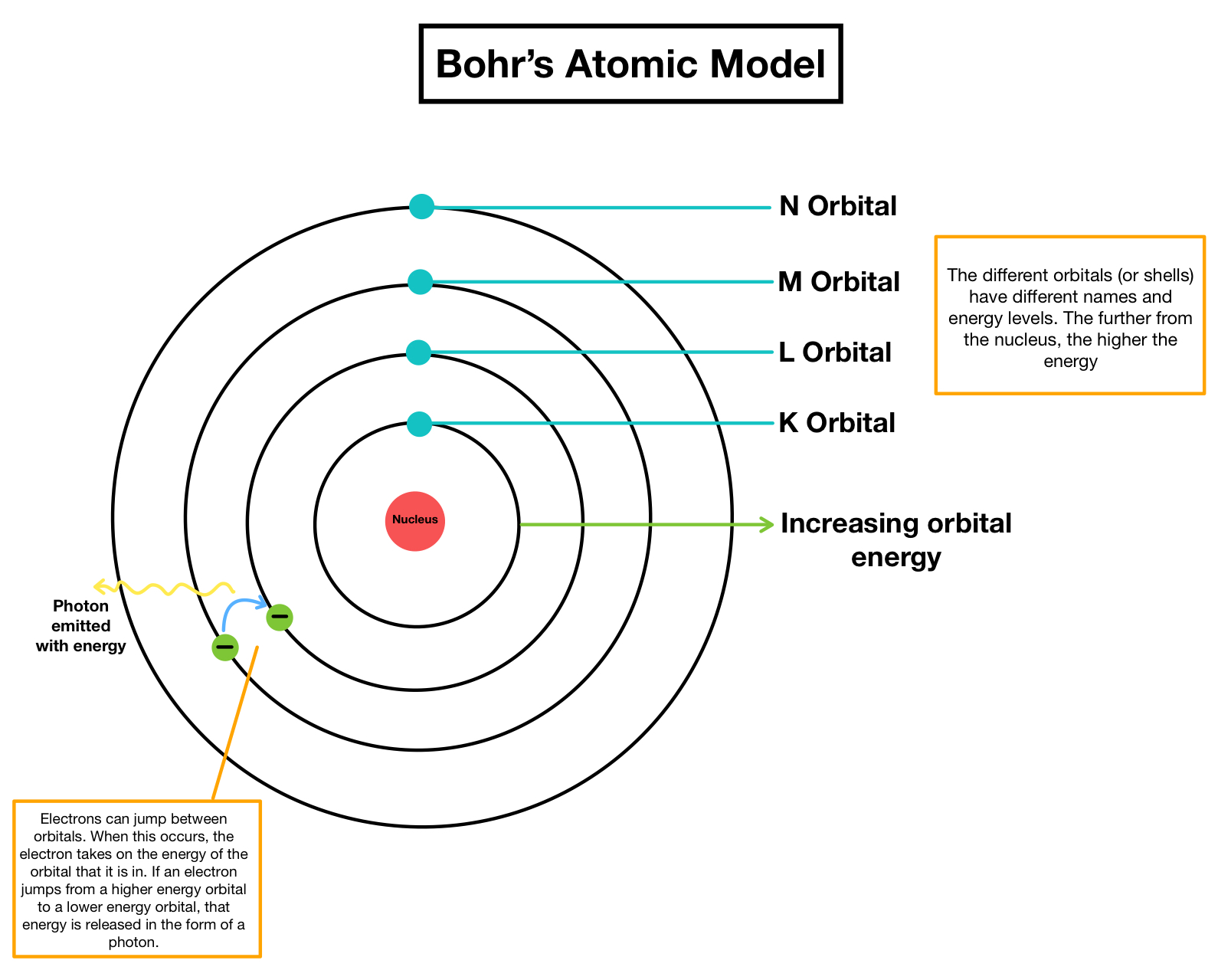
He was born in Illinois in 1868 and later attended and. He found that the ratio of energy in electrons and the frequency of their orbits around the nucleus was equal to. Robert Millikan was an American scientist who made many important contributions to our understanding of atomic theory and electric charge. Atoms of different elements differ in size, mass, and other properties. Atoms of a given element are identical in size, mass, and other properties. Atoms cannot be subdivided, created, or destroyed. Rutherfords gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. The general tenets of this theory are: All matter is composed of extremely small particles called atoms. Scientists had now established that the atom was not indivisible as Dalton had believed, and due to the work of Thomson, Millikan, and others, the charge and mass of the negative, subatomic particles-the electrons-were known. Lord Rutherford pioneered the orbital theory of the atom with his famous gold foil experiment, through which he discovered Rutherford scattering off the nucleus. Bohr turned to Plancks quantum theory to explain the stability of most atoms. Dalton proposed his atomic theory in 1804.
